WAYNE, Pa., June 4, 2018 /PRNewswire/ — Camber Spine today announced that it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market its ENZA™-A Titanium Anterior Lumbar Interbody Fusion (ALIF) system, a unique, minimally invasive interbody fusion device providing integrated fixation.
Daniel Pontecorvo, CEO, said, “ENZA™-A leverages two of Camber Spine’s highly innovative platform technologies. The vast interest and success of our first generation ENZA™ (PEEK) with Duo Presa™ (Two Grips) anchor technology and our line of SPIRA™ products with Surface By Design™, inspired us to combine their defining attributes to create this next generation ENZA™-A Titanium ALIF.”
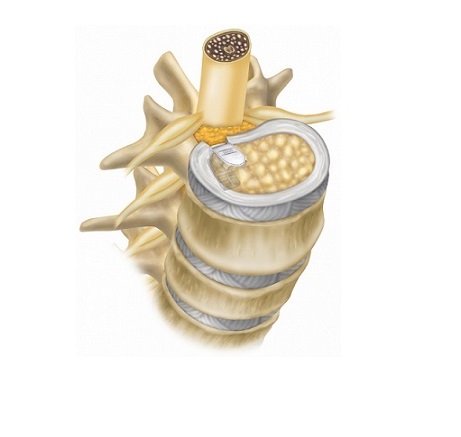
ENZA™-A Titanium ALIF is an interbody fusion device consisting of a 3D printed titanium body with a roughened surface that encourages bone growth onto the cranial and caudal surfaces of the device. The upper and lower faces are deliberately designed with pores that average 500 microns in diameter, the optimal environment for bone to grow and fully incorporate the implant with the vertebral bodies. The ENZA™-A Titanium ALIF has multiple openings to allow a large volume of autogenous bone graft to be easily packed into the implant to further facilitate fusion. Similar to the original ENZA™, this device features two sharpened anchor plates that are housed within the 3D-printed body until they are deployed into the adjacent vertebrae to provide fixation.
According to Seth Anderson, Executive Vice President of New Business Development and Surgeon Relations, “ENZA™-A Titanium ALIF was designed with the surgeon, O.R. staff, and safety in mind. Surgery time is reduced by the single, inline instrumentation used to insert the device, deploy the anchor plates, and lock it in place. Patient safety is increased by minimizing the size of the incision and retraction required for implantation of the device. With ergonomic instrumentation, this system is easy to use and makes implantation more streamlined. The ENZA™-A is the second device in the Camber Spine ENZA™-line of implants. This interbody, coupled with additional product launches expected later this year in the cervical and lateral markets, will continue to grow Camber Spine’s presence as a market leader and innovator in minimally invasive spine surgery technology advancements.”
The Camber Spine ENZA™-A Titanium ALIF is indicated for use with autogenous bone graft in patients with degenerative disc disease (DDD) at one or two contiguous levels from L2 to S1. It is intended to be used with additional FDA-cleared supplementary fixation systems. These implants may be implanted via a laparoscopic or an open anterior approach.
About Camber Spine
WHERE OTHERS SEE PROBLEMS, WE SEE OPPORTUNITIES. TRANSFORMING SURGEON FRUSTRATIONS INTO INNOVATIONS.
Camber Spine, LLP, is a fast-growing musculoskeletal implant company founded in 2010 bringing innovative, best-in-class products to the market, providing surgeons and their patients with better treatment options. The company is committed to delivering surgeon inspired new technologies to the spine market. Camber is an ISO 13485 certified medical device company. Camber Spine Technologies, located in Wayne, Pennsylvania, markets a line of proprietary musculoskeletal products nationwide through its exclusive distributor, S1 Spine. For further information please visit www.cambermedtech.com. For inquiries about ENZA™-A Titanium ALIF or distribution opportunities please call 484.427.7060.
All of Camber Spine Technologies’ products are proudly MADE IN THE USA.
SOURCE Camber Spine