18 December 2018
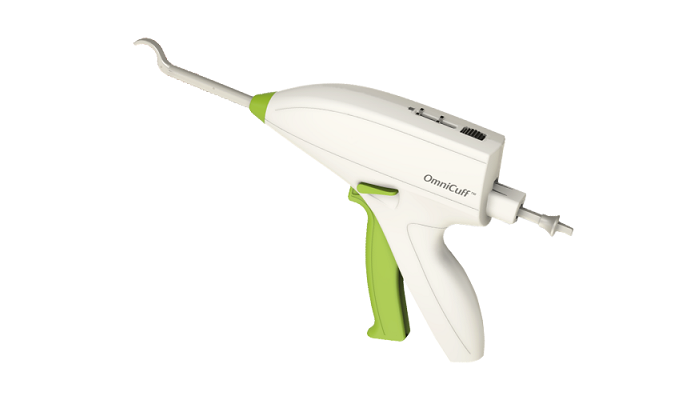
Smith & Nephew plc (LSE:SN, NYSE:SNN), the global medical technology business, today announces that it has agreed to acquire Ceterix® Orthopaedics, Inc, the developer of the NovoStitch® Pro Meniscal Repair System.
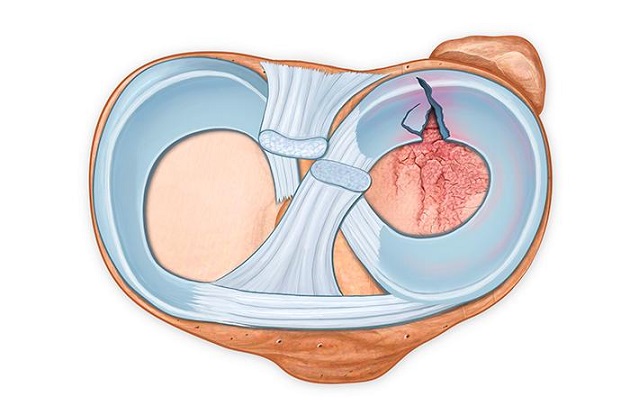
This unique device addresses complex meniscal tear patterns not adequately served by other repair systems. It is highly complementary to Smith & Nephew’s leading FAST-FIX◊ 360 Meniscal Repair System, which addresses vertical tears, the most commonly repairable meniscal injury today.
Currently more than 1.2 million1 meniscal tears are treated surgically in the US each year. In only 15-20%1 of the cases is the meniscus repaired, rather than removed. With products like NovoStitch Pro and FAST-FIX 360 we see the opportunity to double this proportion in the medium term.
Smith & Nephew will pay an initial cash consideration of $50 million and up to a further $55 million over the next five years, contingent on financial performance.
“NovoStitch Pro is an outstanding technology that addresses an unmet clinical need,” said Brad Cannon, President Sports Medicine and ENT, Smith & Nephew. “We are excited by the opportunities to take this new option to our customers. No other company is better positioned to support changing clinical practice as the standard for meniscal treatment pivots from resection to repair.”
The NovoStitch Pro allows surgeons to repair arthroscopically a broader range of meniscal tear types, including horizontal, radial, complex, bucket handle and root tears, as well as vertical tears.
“The new NovoStitch Pro Meniscal Repair System is one of the most innovative technologies developed for arthroscopic knee repair,” said Dr. Peter Kurzweil, president of Memorial Orthopaedic Surgical Group in Long Beach, California. “NovoStitch offers the potential to repair tear types that were previously considered difficult or impossible to sew, with good control and access for the surgeon.”
The NovoStitch Pro will be sold through Smith & Nephew’s extensive sports medicine sales force as well as Ceterix’s existing dedicated sales force. NovoStitch Pro has 510(k) clearance for sale in the US.
“We are proud of the impact our technology has made in developing the meniscal repair market and are excited by the opportunity to reach many more customers and their patients as an integrated part of Smith & Nephew’s extensive Sports Medicine portfolio,” said John McCutcheon, Chief Executive Officer of Ceterix.
The transaction is expected to close in early 2019, subject to the satisfaction of customary conditions. The acquisition will be financed from existing cash and debt facilities.
Investors
Andrew Swift
+44 (0) 20 7960 2285
Smith & Nephew
Media
Charles Reynolds
+44 (0) 1923 477314
Smith & Nephew
Ben Atwell/ Andrew Ward
+44 (0) 20 3727 1000
FTI Strategic Consulting
About Smith & Nephew
Smith & Nephew is a global medical technology business dedicated to supporting healthcare professionals in their daily efforts to improve the lives of their patients. With leadership positions in Orthopaedic Reconstruction, Advanced Wound Management, Sports Medicine and Trauma & Extremities, Smith & Nephew has more than 15,000 employees and a presence in more than 100 countries. Annual sales in 2017 were almost $4.8 billion. Smith & Nephew is a member of the FTSE 100 (LSE:SN, NYSE:SNN).
For more information about Smith & Nephew, please visit our website www.smith-nephew.com, follow @SmithNephewplc on Twitter or visit SmithNephewplc on Facebook.com.
References
1. 2018 SmartTRAK US Meniscal Repair Fixation market report
Forward-looking Statements
This document may contain forward-looking statements that may or may not prove accurate. For example, statements regarding expected revenue growth and trading margins, market trends and our product pipeline are forward-looking statements. Phrases such as “aim”, “plan”, “intend”, “anticipate”, “well-placed”, “believe”, “estimate”, “expect”, “target”, “consider” and similar expressions are generally intended to identify forward-looking statements. Forward-looking statements involve known and unknown risks, uncertainties and other important factors that could cause actual results to differ materially from what is expressed or implied by the statements. For Smith & Nephew, these factors include: economic and financial conditions in the markets we serve, especially those affecting health care providers, payers and customers; price levels for established and innovative medical devices; developments in medical technology; regulatory approvals, reimbursement decisions or other government actions; product defects or recalls or other problems with quality management systems or failure to comply with related regulations; litigation relating to patent or other claims; legal compliance risks and related investigative, remedial or enforcement actions; disruption to our supply chain or operations or those of our suppliers; competition for qualified personnel; strategic actions, including acquisitions and dispositions, our success in performing due diligence, valuing and integrating acquired businesses; disruption that may result from transactions or other changes we make in our business plans or organisation to adapt to market developments; and numerous other matters that affect us or our markets, including those of a political, economic, business, competitive or reputational nature. Please refer to the documents that Smith & Nephew has filed with the U.S. Securities and Exchange Commission under the U.S. Securities Exchange Act of 1934, as amended, including Smith & Nephew’s most recent annual report on Form 20-F, for a discussion of certain of these factors. Any forward-looking statement is based on information available to Smith & Nephew as of the date of the statement. All written or oral forward-looking statements attributable to Smith & Nephew are qualified by this caution. Smith & Nephew does not undertake any obligation to update or revise any forward-looking statement to reflect any change in circumstances or in Smith & Nephew’s expectations.
◊ Trademark of Smith & Nephew. Certain marks registered US Patent and Trademark Office.