Memphis, Tenn., (November 27, 2018) – In2Bones Global, Inc. today announces the commercial launch of the CoLink® View Plating System.
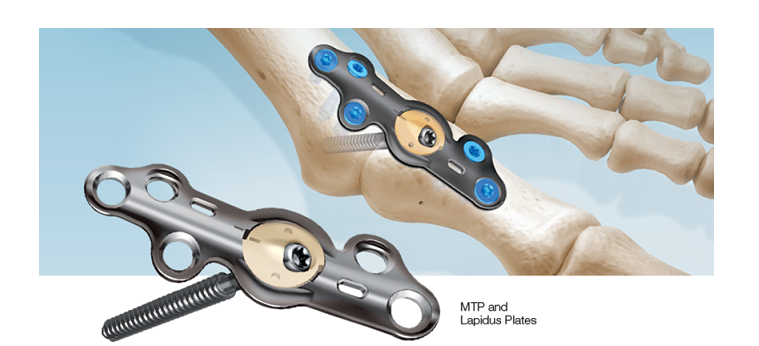
CoLink® View Bone Plates, marketed under the CoLink® View brand name, incorporate a unique combination of titanium metal and a high-performance polymer called polyether ether ketone (PEEK).
The CoLink® View Plates offer transverse joint compression through an X-ray transparent PEEK hub. This allows the surgeon to watch the patient’s bone fusion site during the post-surgical healing process.
Traditional metal plates cannot do this because metal absorbs X-rays and blocks the image underneath. The “see-through” hub is unique to this new In2Bones plating system, and one of many device innovations designed by the company.
In addition to X-ray transparency, PEEK has a benefit of being less stiff than metal and more similar to the elasticity of bone. Bones need mechanical stress (elasticity) during the healing process. Stiffer, all metal implants may eliminate the positive elasticity and slow healing.
The CoLink® View is the sixth In2Bones PEEK-related implant system to reach commercialization. It demonstrates In2Bones’ commitment to expanding the extremities market by launching innovative technological advancements to improve surgical outcomes and patient care.
For more information about In2Bones Global, Inc. extremity products, please visit www.In2Bones.com.
About In2Bones
Headquartered in Memphis, Tennessee, In2Bones is a global designer, manufacturer and distributor of medical devices for the treatment of disorders and injuries of the upper (hand, wrist, elbow) and lower (foot and ankle) extremities. For more information, visit www.i2b-usa.com.
Safe-Harbor Statement:
This press release contains “forward-looking statements” within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. All statements made in this press release, other than statements of historical fact, are forward-looking statements. Forward-looking statements reflect management’s current view of future performance, results, and trends and may be identified by their use of terms such as “anticipate,” “believe,” “could,” “estimate,” “expect,” “intend,” “may,” “plan,” “predict,” “will,” and other similar terms. The company wishes to caution readers that actual results might differ materially from those described in the forward-looking statements. Forward-looking statements are subject to a number of risks and uncertainties impacting the business, including increased competition, technical obsolescence, regulatory issues, general economic conditions and other risks.
Media Contacts:
Jon Simon | Sr. Vice President of Marketing
In2Bones USA
6000 Poplar Ave, Suite 115 Memphis, TN 38119
M: 512.739.5793 • O: 901.260.7931 • F: 844-712-6637
Email: jsimon@i2b-usa.com
https://www.linkedin.com/company/22344175/
Bob Reed
Element-R Partners
630.579.1770
bob@rurelevant.com