March 1, 2017
BORDEAUX, France & BOSTON–(BUSINESS WIRE)–
IMPLANET (IMPL.PA) (IMPZY) (Euronext: IMPL, FR0010458729, PEA-PME eligible; OTCQX: IMPZY), a medical technology company specializing in vertebral and knee-surgery implants, announces that the French patent office has granted a patent protecting the JAZZ Lock® implant in France.
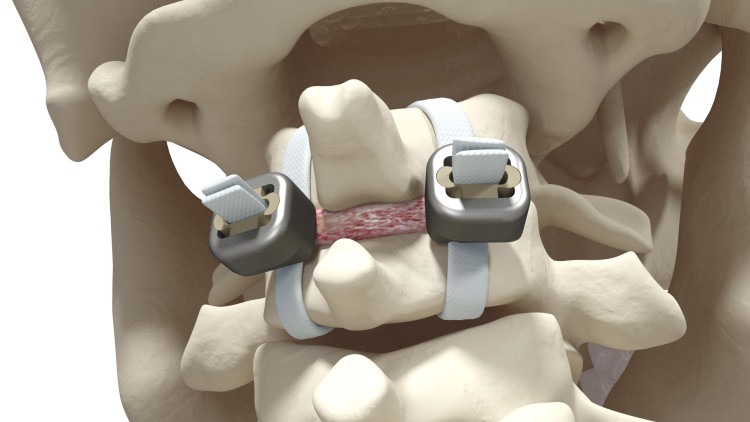
A major component of an innovative range of band products for spine surgery, Jazz Lock® is an implant designed to treat degenerative spine disorders, whose global market potential is estimated at over $200 million.
An unrivaled implant, thanks to its locking system requiring no rod fixation, Jazz Lock® allows surgeons to shorten and simplify the surgical procedure by removing multiple steps and implants used in traditional system: locking screws and connecting rods.
Régis Le Couedic, Implanet’s Product Development & Manufacturing Director, says: “Obtaining this patent from the French patent office is the first step in fully protecting our Jazz Lock®, and more particularly, its locking mechanism that is the first of its kind in the market. The protection of this latter element is pivotal, insofar as the majority of our future developments will incorporate this technology. We continue to be extremely rigorous regarding the protection of our current and future innovations.”
Ludovic Lastennet, CEO of Implanet, adds: “We are continuing the strict execution of our development strategy, of which the comprehensive protection of our products is a major component. We have submitted the dossier to various global intellectual property authorities, notably the United States. Since its limited launch last autumn in the United States and Europe, Implanet has sold more than 300 Jazz Lock® implants to treat degenerative spine disorders. The clinical feedback from surgeons has been excellent, including important practical aspects such as ease of use and speed of implementation.”
Next financial press release: 2016 annual results, on March 28, 2017
IMPLANET will participate in the following events:
- GES (“Groupement d’étude sur la Scoliose” scoliosis study group), Strasbourg, France, March 9 to 11, 2017
- AAOS (American Academy of Orthopedic Surgery), San Diego CA, USA, March 15 to 17, 2017
About IMPLANET
Founded in 2007, IMPLANET is a medical technology company that manufactures high-quality implants for orthopedic surgery. Its flagship product, the JAZZ latest-generation implant, aims to treat spinal pathologies requiring vertebral fusion surgery. Protected by four families of international patents, JAZZ has obtained 510(k) regulatory clearance from the Food and Drug Administration (FDA) in the United States and the CE mark. IMPLANET employs 48 staff and recorded 2016 sales of €7.8 million. For further information, please visit www.implanet.com.
Based near Bordeaux in France, IMPLANET established a US subsidiary in Boston in 2013.
IMPLANET is listed on Compartment C of the Euronext™ regulated market in Paris.
View source version on businesswire.com: http://www.businesswire.com/news/home/20170228007033/en/