ENGLEWOOD, Colo., Jan. 30, 2017 /PRNewswire/ — Since its inception, Paragon 28 has obsessed over every aspect of foot and ankle surgery. Committed to creating tailored solutions to improve surgical outcomes, Paragon 28 has launched innovative products and instrumentation that help to streamline procedures, allow surgeons flexibility in technique and approach, and facilitate reproducible results benefitting both the surgeon and patient.
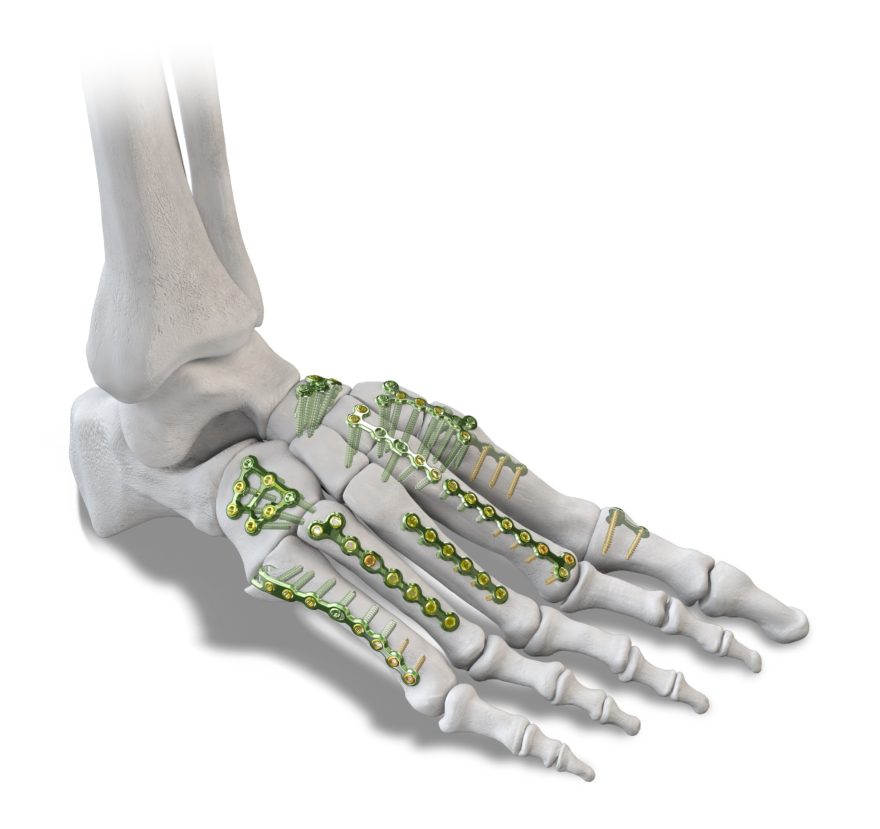
Today, Paragon 28 continues to honor a tradition of comprehensive and innovative solutions with the launch and release of the Baby Gorilla® Mini Plating System.
The Baby Gorilla® Mini Plating System leverages the modularity, optionality and specificity admired in the classic Gorilla® Plating system and offers a comprehensive set of implants for procedures that may require narrower plates and smaller screws matching the size and anatomy of small bones within the foot and ankle.
The system provides 65 unique Baby Gorilla® plating options from 13 plating families. All plates are designed to accept both 2.0 mm and 2.5 mm locking and non-locking variable angle plate screws.
The system offers the most extensive array of universal utility, fracture, and procedure specific plates which require minimal manipulation to achieve satisfactory results.
Plate options found within the Baby Gorilla® Plating System include:
The Baby Gorilla® Mini Plating System also includes a robust assortment of specially designed instrumentation including curettes, reduction clamps, osteotomes, plate bending tools and compression and distraction devices engineered specifically for use in the foot and ankle for small bone fractures and reconstruction procedures.
Paragon 28 is grateful for the significant contributions Dr. Patrick Yoon, MD Hennepin County Medical Center Minneapolis, MN and Dr. Thomas Harris, MD UCLA-Harbor Medical Center, Torrance, CA made as surgeon designers of this system.
About Paragon 28, Inc.
Paragon 28, Inc. was established in 2010 to address the unmet and under-served needs of the foot and ankle community. From the onset, Paragon 28 has made it our goal to re-invent the space of foot and ankle surgery. We believe that through research and innovation we can create new and improved solutions to the challenges faced by foot and ankle specialists.
Product Page:
http://www.paragon28.com/by-system/baby-gorilla/
SOURCE Paragon 28, Inc.