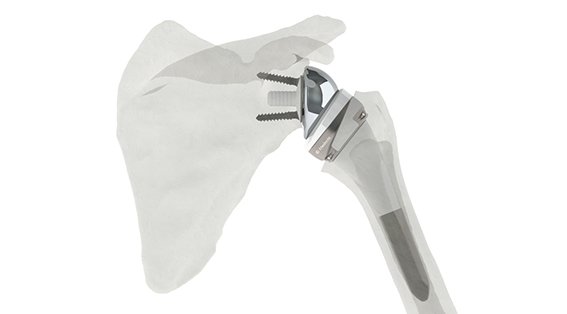
TAMPA, Fla., Feb. 22, 2017 /PRNewswire/ — Corin Group announced today the launch of the Humelock Reversed Shoulder System, a device used to treat patients with both a massive rotator cuff tear and a severe form of shoulder arthritis. The condition, known as cuff tear arthropathy, causes pain and reduced mobility in the arm and shoulder.
The Humelock Reversed Shoulder System, which was developed by FX Solutions, a privately-held upper extremities technology company based near Lyon, France, received FDA clearance for the device in January 2017. The device is already available in Europe and other international markets.
Corin Group holds a distribution agreement for the device, as well as the Humelock II™ Cementless and Cemented Anatomical and Reversible Fracture Shoulder Systems. The agreement includes distribution rights in the United States, Germany and the United Kingdom.
“Our partnership with FX Solutions allows Corin to increase their offering in shoulder arthroplasty, whilst we continue to expand solutions in hip and knee. Our goal is to provide surgeons, hospitals, health systems, and patients with a new and comprehensive approach to orthopaedics and joint replacement,” said Paul Berman, President, Corin USA. “The launch of the Humelock Reversed Shoulder System means patients in the U.S. will have access to an important new option for reverse shoulder replacement.”
“Our continuing partnership with Corin provides U.S. clinicians access to our surgeon-designed shoulder systems, which have demonstrated clinical success in European markets,” said FX Solutions’ President and CEO Jean-Jacques Martin. “Corin is a valued partner committed to innovation that improves health outcomes and increases both patient and surgeon satisfaction. We look forward to continued success together.”
About Corin Group
Corin is a European orthopaedic manufacturer based in the UK that markets its products throughout the world.
Corin is committed to:
…improving patient satisfaction with personalized technologies that optimize our clinically proven joint replacements
…delivering a personal approach to our customers, combining the spirit of our local companies with the strength of our global, integrated organization
…empowering and rewarding our global talented teams to deliver excellence to our customers
For further information about Corin, please visit www.coringroup.com.
This news release contains forward-looking statements. These statements appear in a number of places in this news release and include statements regarding our intentions, beliefs or current expectations, concerning, among other things, our results of operations, turnover, financial condition, liquidity, prospects, growth, strategies, new products, the level of new launches and the markets in which we operate. Readers are cautioned that any such forward-looking statements are not guarantees of future performance and involve risks and uncertainties, and that actual results may differ markedly from those in the forward-looking statements as a result of various factors. We undertake no obligation publicly to revise any forward-looking statements, except as may be required by law.
SOURCE Corin USA